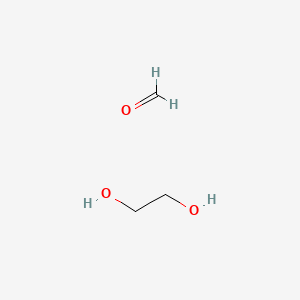
Crystalline ethane-1,2-diol does not have intra-molecular hydrogen bonding: Experimental and theoretical charge density studies - ScienceDirect

Which is the most stable conformer of ethane-1,2-diol?A)\n \n \n \n \n B)\n \n \n \n \n C)\n \n \n \n \n D)\n \n \n \n \n

Ethylene glycol (ethane-1,2-diol), technically at least 99.5 % | Ethylene glycol (ethane-1,2-diol), technically at least 99.5 % | Coolants | chemiekontor.de
Which is most stable conformer of ethan-1,2-diol - Sarthaks eConnect | Largest Online Education Community

Ethane - 1,2 - diol has the given structure.Without breaking the C - C bond, there are five possible oxidation products.What is the total number of aldehyde groups and carboxylic acid groups

Aerobic oxidative cleavage of 1,2-diols catalyzed by atomic-scale cobalt-based heterogeneous catalyst | Communications Chemistry