The pH of an acetic acid solution is 3.26. What is the concentration of acetic acid and what is the percent of acid that's ionized? - Quora

The dissociation constants for acetic acid and HCN at 25C are 1.5 x 10^-5 and 4.5 x 10^-10 respectively. The equilibrium constant for the equilibrium CN + CH3COOH HCN + CH3COO - would be ?
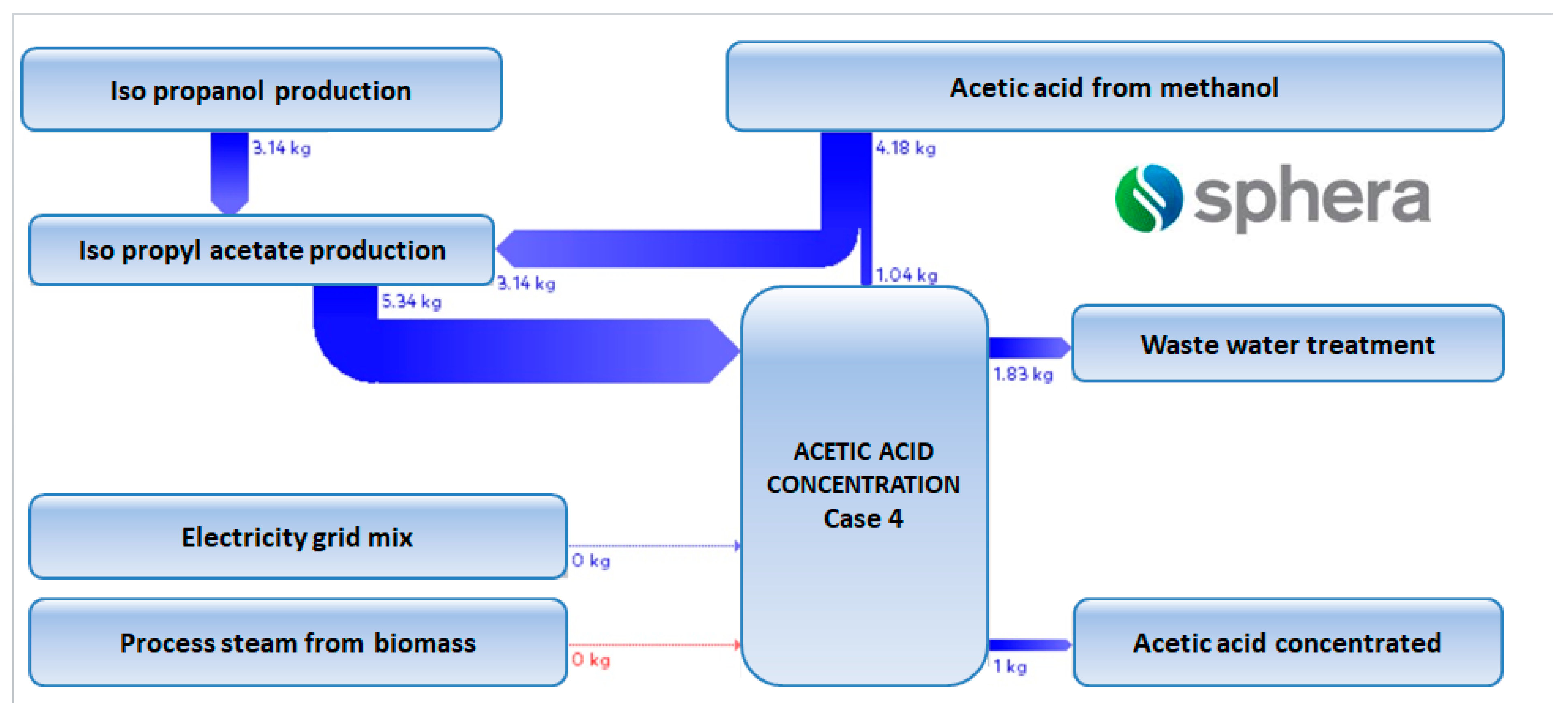
Energies | Free Full-Text | Classical and Process Intensification Methods for Acetic Acid Concentration: Technical and Environmental Assessment

Calculate t the `H^(+)` ion concentration in 0.10 M acetic acid solution. Given that the dissociati - YouTube

Dilmeet Sawhney- Determination of Acetic Acid in Vinegar Lab Report. Parts 1 & 2.docx - Determination of Acetic Acid Concentration in Vinegar Lab | Course Hero

The ionisation constant for acetic acid is 1.8xx10^(-5). At what concentration will it be dissociated to 2% ?
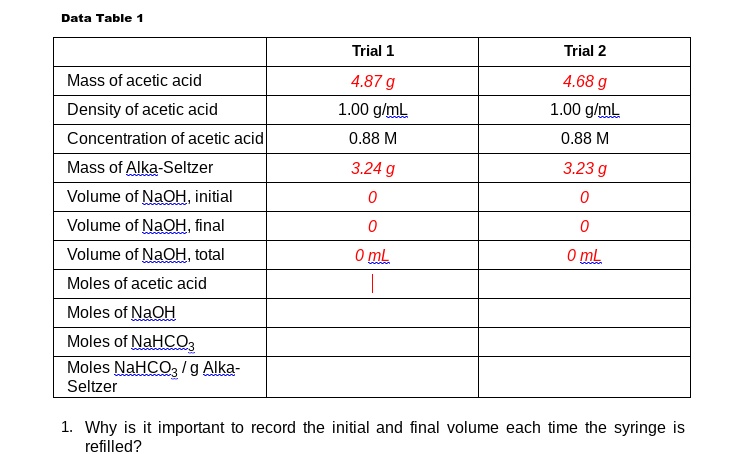
SOLVED: Data Table Trial 1 Trial 2 Mass of acetic acid Density of acetic acid Concentration of acetic acid Mass of Alka-Seltzer Volume of NaQH, initial Volume of NaQH; final Volume of



![PDF] Influence of acetic acid concentration on the solubilization of chitosan | Semantic Scholar PDF] Influence of acetic acid concentration on the solubilization of chitosan | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/d4193be5618526a91bb40f65dc033b480dcfce4d/2-Figure1-1.png)